![]()
Discovery of Silicon
In 1787, the French chemist Antoine-Laurent de Lavoisier first discovered the silicon present in rocks. In 1800, silicon was mistaken by Sir Humphry Davy as a compound. In 1811, French chemists Joseph Louis Gay-Lussac and Louis Jacques Thénard probably prepared impure amorphous Silicon by heating potassium with silicon tetrafluoride. They later named it silicon according to the Latin silex (meteorite).
Until 1823, silicon was first discovered in the form of a metal element by the Swedish chemist Jöns Jacob Berzelius. One year later, he extracted amorphous silicon in much the same way as Gay-Lussac, and then purified the elemental silicon by repeated cleaning; in the same year, he heated the silicon oxide powder and the mixture of iron and carbon at a high temperature and obtain the iron silicide.
In order to extract pure silicon, Berzelius dry-fired the silicon-fluorine-calcium compound, hydrolyzed the obtained solid, and manage to obtain the pure silicon. In 1824, in Stockholm, Berzelius obtained relatively pure silicon powder by heating potassium fluorosilicate and potassium. Therefore, it is agreed that the honor of discovering silicon belongs to Berzelius.
Properties of Silicon
| Symbol: | Si |
| Atomic Number: | 14 |
| Atomic Weight: | 28.09 |
| Element Category: | metalloid |
| Color: | dark gray with a bluish tinge |
| Density: | 2328.3 kg/m³ |
| Hardness: | 6.5 |
| Proportion in Earth’s Crust: | 25.7% |
| Other Names: | Silicium, Silicio |
Application of Silicon
High-purity monocrystalline silicon is an important semiconductor material that can be used as a solar cell to convert radiant energy into electrical energy, which is a promising material in the development of energy.
Silicon can also be made into cermet composites, which are resistant to high temperatures, toughness, and can be cut. They not only inherit the respective advantages of metals and ceramics, but also make up for the inherent defects of both, and can be applied to weapons manufacturing and aerospace.
Pure silica can be used to draw high transparency glass fiber for optical fiber communication, which is the latest modern communication means.
Please visit https://www.sputtertargets.net/ for more information.


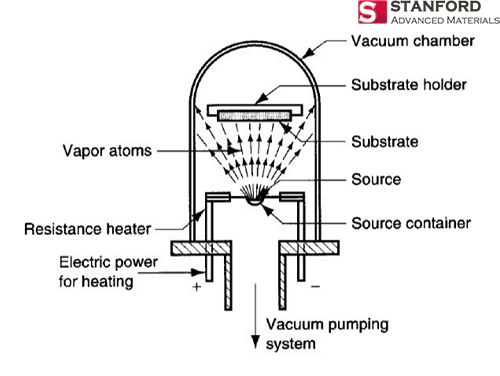
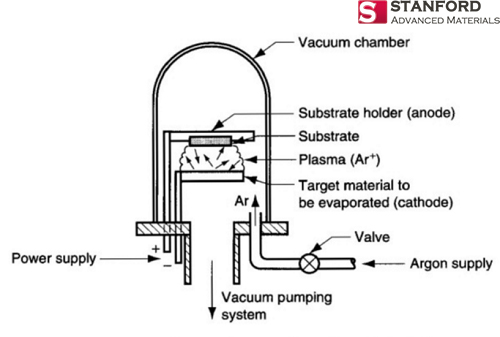
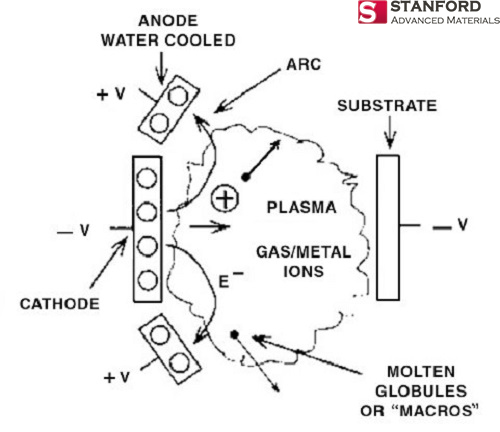



 Cyprus is the original source material for the chemical element Copper. The people of Middle East initially discovered it in 9000 BC. “Cu” is the canonical chemical symbol of copper.
Cyprus is the original source material for the chemical element Copper. The people of Middle East initially discovered it in 9000 BC. “Cu” is the canonical chemical symbol of copper. Whereas Sulfur, otherwise known as sulphur, is first introduced in 2000 BC and discovered by Chinese and Indians. It is a chemical name originated from the Sanskrit word ‘sulvere’, and the Latin ‘sulfurium’. Both names are for sulfur.
Whereas Sulfur, otherwise known as sulphur, is first introduced in 2000 BC and discovered by Chinese and Indians. It is a chemical name originated from the Sanskrit word ‘sulvere’, and the Latin ‘sulfurium’. Both names are for sulfur.








